Company Updates
Keep Reading 

GVD Braille Height Sets A New Standard for Precision and Speed in Braille Inspections for EU Pharmaceutical Market
Date: April, 2024 | Category: Compliance | Author: Hana Trokic GlobalVision’s Braille Height is breaking new ground by offering unmatched precision and speed never before seen for regulatory Braille inspections. With the new GVD…

The Verify x Esko WebCenter Integration Automates Proofreading and Inspections for Packaging Artwork
Date: March, 2024 | Category: Proofreading | Author: Hana Trokic The Verify x Esko WebCenter Integration represents a digital shift in the packaging artwork review process.
Verify x Esko WebCenter - An Integration to Unify Your…

Verify’s New AI-Powered OCR Feature Brings Unmatched Character-for-Character Inspections to Regulated Industries
Date: February, 2024 | Category: Company | Author: Hana Trokic Verify's New AI-Powered OCR Feature is Here! Verify, GlobalVision’s newest and most innovative cloud-based proofreading software, has just raised the bar for proofreading…
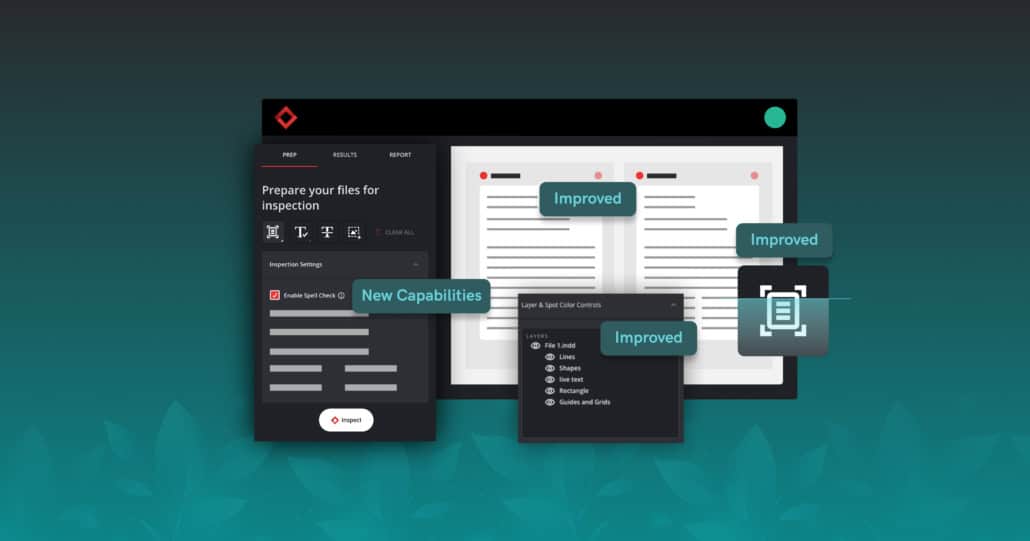
Proofreading Has Never Been Easier – Verify’s Winter Release Brings a New Approval Workflow for Custom Dictionary Words and More
Introducing our Verify Winter Release, an update that brings enhanced functionalities designed to elevate your compliance review experience by redefining precision in automated proofreading. With new versatile capabilities, this update broadens the scope of applicable use cases, amplifies workflow efficiency, and elevates the accuracy of your inspection results.

Revolutionizing Braille Inspection: Introducing the Upgraded GVD Braille Module
From a quality control perspective, Braille Inspection and accuracy play an important role as Braille is mandatory on pharmaceutical packaging all across Europe, and is strongly recommended by the FDA in Western markets, while other regions such as the ASEAN markets are working to heighten inclusivity by implementing braille requirements on packaging as well.

Enhancing Your Verify Experience – Our Fall Release is Here
This Verify release marks a huge milestone for Verify’s core technology, as it has been completely rehauled and updated to an entirely new architecture with all modern technology—get ready to elevate your compliance review and inspection processes!

GlobalVision is Going to RAPS Convergence 2023 in Montreal
Join us at one of the top life sciences events of the year as we engage with industry leaders and professionals, highlighting our innovative proofreading solutions while fostering valuable connections. Make sure to stop by our booth #225 during the event to gain valuable insights into our products and services and see what’s new with GlobalVision!

IPG Health Increases Efficiency in Document Reviews With GlobalVision
IPG Health is a network of world-renowned agencies focused on health communication and marketing. Collectively, the network is made up of over 45 agencies that are spread across six continents and 6,500+ employees driven by a company-wide obsession with harnessing creativity, technology, science, and data to inspire behaviors that fuel better health.

Introducing Text Inspection 2: The Next Generation of Compliance Proofreading
We are thrilled to announce the launch of our latest cloud-based proofreading technology, Text Inspection 2. Through significant innovation and constant improvements, our newest algorithm makes inspections 10-30 times faster than before.

Verify x Veeva Vault: Empowering Promotional, Regulatory, and Labeling Teams in Life Sciences to Maximize Content Efficiency
GlobalVision provides life sciences teams with accuracy and efficiency while minimizing the resources required in the review process. Now, with a Veeva Vault RIM Suite integration, highly regulated businesses can achieve efficiency in their review and regulatory workflows while also achieving global efficiency alignment and cross-functional unification — otherwise challenging tasks for large multinational enterprise companies.
Ready for a New Verify Release? 2023.2 is Here
With the latest Verify 2023.2 release, users can expect a heightened and more streamlined automated proofreading experience. This upgraded version comes equipped with new features and capabilities that not only enhance the user experience but also improve the accuracy and efficiency of quality inspections.

GlobalVision Chosen as One of Canada’s Best Places to Work
In recent years, GlobalVision has made extensive efforts to make our organization an amazing place to work and our efforts have not gone unnoticed. In addition to gaining the Great Place to Work certification, this year, we are extremely proud to announce that we won the Best Workplaces in Canada for 2023 award.

Don’t Miss The Proof – GlobalVision’s First Proofreading & Quality Control Digital Conference
We are extremely excited to announce that we are hosting an exclusive online event, specifically designed for proofreading and regulatory professionals in the Pharmaceutical, Life Sciences, and CPG industries. Our digital event will feature a variety of interactive sessions including Ask-an-Expert Q&A, a Fireside Chat, and expert sessions with speakers from our own GlobalVision team, Biogen, Alcon, Gilead, and more.

Join GlobalVision at the Veeva R&D Summit
With live events back in full swing this year, GlobalVision is taking notes and taking full advantage. We’re excited to be face-to-face with some of pharma’s top leaders and professionals by taking part in this year’s Veeva R&D and Quality Summit.

How Johnson & Johnson’s Labeling Team Scaled their Quality Review Process
For over 20 years, GlobalVision has been helping Johnson & Johnson teams globally through the power of automation technology. Enabling automated quality checks throughout their packaging workflows is the backbone that has made this partnership so fruitful and successful. From the Consumer Health division to their internal Medical Device giant Ethicon, the nature of J&J’s regulated products makes the need for error-free packaging components an absolute necessity.

GlobalVision Advances to the Silver Certified Level of Veeva’s Technology Partner Program
Proofreading critical content accurately and at scale is a major concern in the Enterprise Pharma and life sciences industry though, currently, content and documentation checks are still mainly done manually. This is otherwise extremely tedious, time-consuming, and with a high margin for human error. Veeva, the leader in cloud-based software solutions for the life sciences industry, and a Veeva Technology partner, GlobalVision, together have found an answer to this problem— an end-to-end solution for regulated industries to scale the creation, review, and distribution of highly critical content.

New Release: GlobalVision Web 3.10
Date: March, 2022 | Category: Company | Author: Hana Trokic Introducing GVW 3.10, GlobalVision’s latest version of the original cloud-based inspection platform. Built with the purpose of enhancing the overall user experience…

Verify 2021.3 New Release
Introducing Verify 2021.3, the latest version of GlobalVision’s web-based inspection application. Built to deliver a more intuitive user experience, Verify optimizes inspections by catching deviations at every revision point.
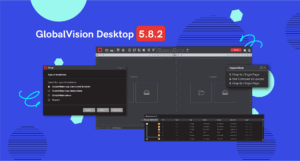
Introducing GlobalVision Desktop 5.8.2
GlobalVision’s latest validated release boasts several exciting features to improve performance and increase stability for desktop users. Desktop version 5.8.2 addresses several bug fixes while providing users with new features to create the most seamless inspection process yet. Notable features in this release include new language preferences for users, UI and Text Grouping features, and a new Installer Update.
Introducing GlobalVision Desktop 5.7: See What’s New
Date: September, 2019 | Category: Company | Author: Kayla Caticchio The latest release from GlobalVision comes with exciting new features to enhance performance for desktop users. Developed with customer feedback in mind, version 5.7 boasts…
GlobalVision Web 3.5 Delivers Custom User Profiles and Improvements to Text & Spelling Inspection
Date: June, 2019 | Category: Company | Author: Kayla Caticchio
The new GlobalVision Web Version 3.5 gives GlobalVision users what they’ve been asking for, delivering several new highly requested features.
In addition to…

Introducing GlobalVision Desktop 5.5: Improved Text & Spelling Inspection and a New Tool for Tobacco Packaging
The latest release from GlobalVision gives desktop users the ability to run faster and more accurate inspections with enhanced Text & Spelling Inspection features. Along with optimized processing speeds and improved usability, Version 5.5 introduces a new add-on tool for the inspection of tobacco packaging.

Access-Level Additions Shape GlobalVision 5.4 into Biggest Desktop Release Yet
Date: February, 2019 | Category: Company | Author: Ryan Szporer <div style="display: none;"> </div><p>Many within the quality-control community should be excited about the latest GlobalVision 5.4 release for desktop.…

GlobalVision Desktop 5.3 and Web 3.3 Deliver Greater User Control
Date: November, 2018 | Category: Company | Author: Ryan Szporer The just-released GlobalVision Desktop 5.3 and GlobalVision Web 3.3 each bring exciting, new features to the forefront. The end result, for both deployment options, is greater…

GlobalVision Adds Electronic Signatures for Enhanced Data Integrity to Most Comprehensive Platform Yet
Date: June, 2018 | Category: Company | Author: Ryan Szporer With regulations increasing exponentially in importance, GlobalVision has responded to customers’ calls for greater control over data integrity by introducing electronic…
Presenting GlobalVision 4.1 for Desktop: New Workflows for Print Production
Having previously pulled out all the stops unifying our apps into a single platform, we’re going further and adding to that all-in-one offering. Presenting our latest desktop release: GlobalVision 4.1. Users can now take advantage of…

Out Now: Artwork Creation Guide Standardization for Digital Files
To catch errors, you first need to see them. Before using GlobalVision to check for mistakes on your files, make sure they’re created the same way. Our Artwork Creation Guide contains insights and easy-to-follow steps to improving…
You’ve Got Questions? The GlobalVision Help Center Has Answers.
You may have already noticed the sleek, revamped look of the GlobalVision website. It’s since extended to our brand-new, easy-to-use Help Center, which provides users with all the know-how they need to get the most out of their…
Why “Web-Enabled” Apps Aren’t Really Web Apps
Date: May, 2017 | Category: Company | Author: Jonathan Hou You see the words “web,” “cloud,” and “internet of things” just about everywhere you go these days. In today's digital age, where "there's an app" for almost everything,…
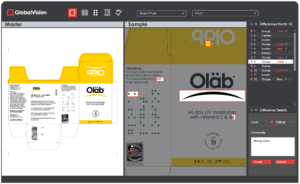
The GlobalVision Platform has Transformed and it’s No Mistake
At GlobalVision, we understand the importance of aiming for perfection. With so much competition across endless industries, one slip up could mean the difference between the success or failure of a business’s quarter for the business. All…
Announcing GlobalVision Technologies Built into Esko Automation Engine
Forget 7. 16 is the next big thing. This week marks the iPhone 7 launch but for us prepress folks, we’ve got something to be more excited about with the launch of Esko Software Platform 16. 16 marks the arrival of GlobalVision’s…

CMOs of Increasing Importance to Pharmaceutical Companies
Date: July, 2016 | Category: Compliance | Author: Reuben Malz The pharmaceutical marketplace has changed dramatically over the last few years as pharmaceutical companies are moving from centralized, internal production to single-source…
Esko Software Platform Makes Packaging Simplified
Date: April, 2016 | Category: Company | Author: Reuben Malz
Ghent (Belgium), March 16, 2016 – Esko has wholly updated its software solutions and relaunched them under the umbrella brand name “Esko Software Platform”. It’s…
![]()
GlobalVision is an intelligent inspection
platform that finds errors in your work
before they become problems.
Stay updated with GlobalVision by signing up for our newsletter.
Contact Us
GlobalVision HQ
16800 Route Trans-Canada
Montreal, QC, Canada
H9H 4M7
+1-514-624-4422
Company
Contact us
About
Partners
News & Events
Careers
Security
Privacy Settings
Subscribe to our newsletter.